A Food and Drug Administration advisory committee voted against approving the booster dose of Pfizer-BioNTech Covid-19 vaccine for most Americans. The vote, which was non-binding, was 16-2, and goes contrary to President Joe Biden’s plans to start booster shots next week.
Biden and his top health adviser Dr. Anthony Fauci have said that booster shots would be needed eight months after a person’s second Pfizer shot.
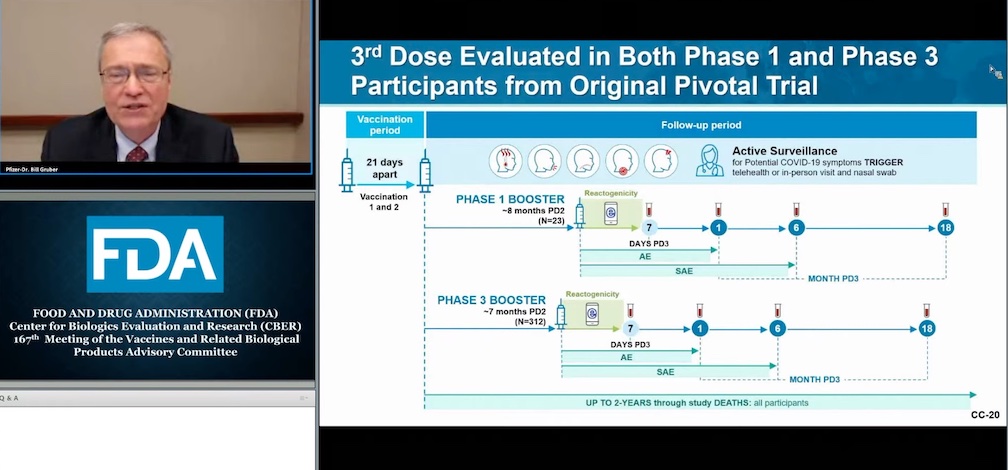
The meeting participants today went through a 23-page FDA presentation where there was a lot of support for a booster shot, but panelist did not agree with some of the presentation conclusions, and said more data is needed before it can recommend the third shot.
The FDA does not need to heed the advice of the panel, however, but so far it has followed this group’s advice concerning vaccines.
“In particular, there is a lack of data on effectiveness and duration,” said Dr. Michael Carome, who serves on the panel and is the director of Public Citizen’s Health Research Group. “Current evidence does not appear to show a need for boosting.”
The advisory panel said that the two initial doses of the Pfizer shot is highly effective against the Delta variant of Covid-19, and said boosters are not needed at this time.
Watch the meeting at this link: